Clinical validation
 Prenatal
Diagnosis
Prenatal
Diagnosis, one of the most prestigious journals in prenatal diagnosis field, has recently published - dedicating the cover - a prospective study conducted by the
GENOMA Group Research Team on the clinical utility of the
PrenatalSafe®
Karyo test.
Aim of the study
The aim of the study was to evaluate the
PrenatalSAFE® Karyo
test clinical utility (which extends the analysis to all chromosomes by
detecting both aneuploidies and structural chromosomal abnormalities)
compared to
traditional NIPTs (which restrict the analysis to only 5
chromosomes: 21, 18, 13, X and Y), and, at the same time, to evaluate the
influence of the increase in sensitivity on specificity. The study was
conducted on clinical samples.
Method and materials
In the study, where recruited
12.114 pregnant women (both
high and
low risk pregnancies) who underwent the non-invasive screening
test of fetal aneuploidies common in pregnancy (Trisomy 21, Trisomy 18,
Trisomy 13 and X and Y sex chromosomes aneuploidies), by analysing
circulating free fetal DNA (cfDNA). Blood samples collected from the
pregnant women were analysed, as well as with a traditional NIPT for the
research of aneuploidy on the 5 chromosomes, even with a genome-wide
technology to detect aneuploidies and structural chromosomal abnormalities
extending the analysis to the entire karyotype, i.e. with the technology
used by the
PrenatalSAFE®
Karyo test.
Obtained results
Il test
PrenatalSAFE® Karyo
test allowed to identify
18/169 (10.7%) pregnancies with fetal chromosomal abnormalities of clinical significance that a traditional NIPT could not detect,
12 (7.4%) of which could have been completed and result in the birth of children with chromosomopathies. Data from the study demonstrate the clinical utility of
PrenatalSAFE® Karyo
test that, compared to traditional NIPTs,
increased the sensitivity from 92.64% to 100% (p<0.001). Furthermore, the results show that despite the increased sensitivity obtained with the screening level of the fetal karyotype,
the test specificity did not decrease significantly, only diverging 0.1% from the specificity of a traditional NIPT
(99.77% vs 99.87%, p=0.064).
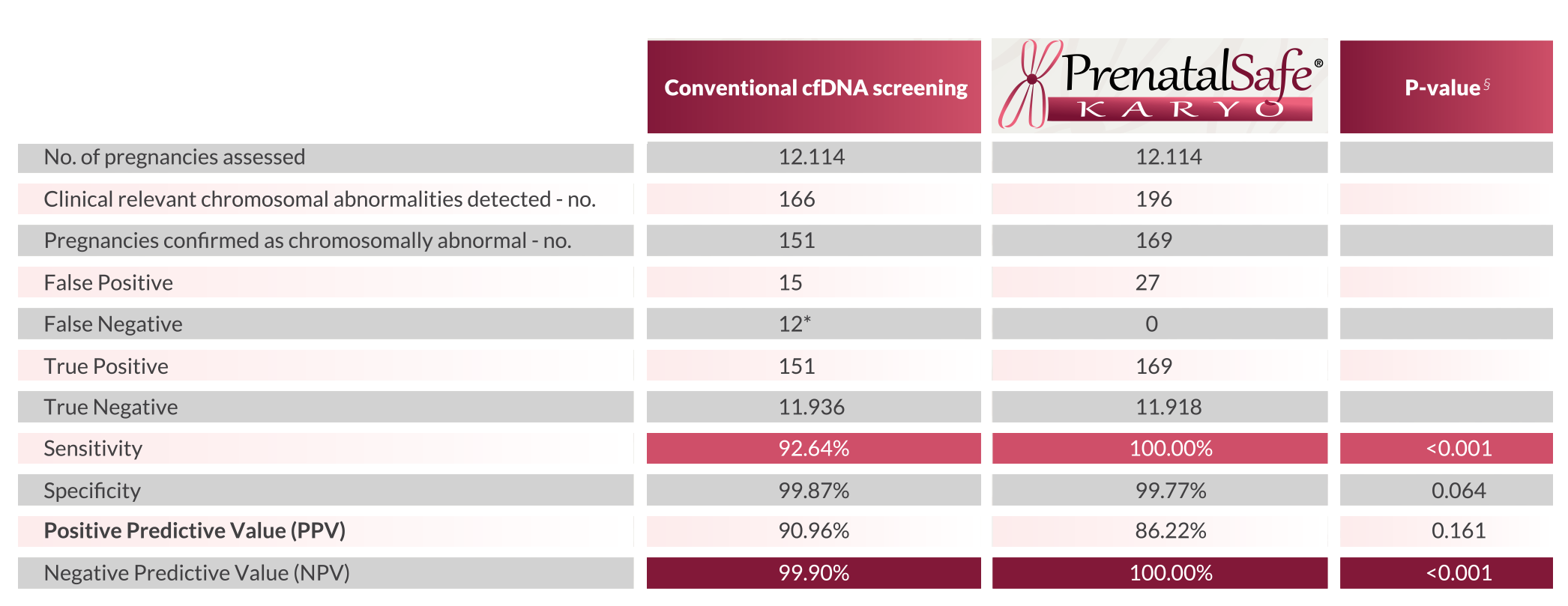 * Clinically relevant chromosomal abnormalities, not detected by conventional cfDNA screening, potentially resulting in the birth of babies with chromosomal anomalies, have been considered as false negative.
§ A P-value of less than 0.05 was considered to indicate statistical significance.
* Clinically relevant chromosomal abnormalities, not detected by conventional cfDNA screening, potentially resulting in the birth of babies with chromosomal anomalies, have been considered as false negative.
§ A P-value of less than 0.05 was considered to indicate statistical significance.
Conclusions
Highlighting
the importance of non-invasive screening of the entire fetal karyotype,
the study confirmed the usefulness of the new method not only to detect
aneuploidies on all fetus’ chromosomes, but especially for the research of
structural abnormalities (deletions and duplications of chromosome
portion), possible only with the latest high-resolution technology, such
as the one used in the
PrenatalSAFE® Karyo test.
Share: